The electron mass has the smallest mass between the basic particles inside the atoms

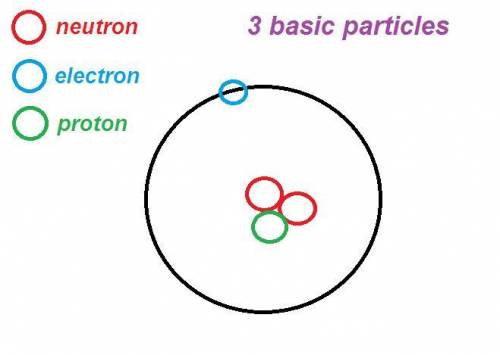
Inside the atom consists of a nucleus containing 3 basic particles namely protons and neutrons, while electrons move around the nucleus.
The charge of 1 proton is equal to a charge of 1 electron, but the sign is different.
The proton is positively charged (+1), the electron is negatively charged (-1). and neutrons not charged (neutral)
For a charge of 1 electron = e = 1.66.10⁻¹⁹ coulomb
Whereas the neutron charge = 0
The mass number is not the same as the atomic mass. The mass number is the number of protons and neutrons in one atom. While the atomic mass is a number that expresses the mass of one atom.
Atomic mass is expressed in special units, namely atomic mass unit because the atomic mass is very small if expressed in units of grams or milligrams
This atomic unit uses the standard atomic mass, that is, the C-12 isotope
1 atom C-12 = 12 atomic mass unit
1 atomic mass unit = 1/12 x mass 1 atom C-12
The mass of 1 proton is equal to the mass of 1 neutron, equal to 1 atomic mass unit and electrons have a mass smaller than both.
electron mass = 9.11.10⁻²⁸proton mass = 1.6726.10⁻²⁴neutron mass = 1,675.10⁻²⁴
The proton mass is considered to be equal to 1 atomic mass unit and a neutron which has a slightly larger mass is also considered to be equal to 1 atomic mass unit
1 unit of atomic mass = 1.6726.10⁻²⁴
So the electron mass has the smallest mass between the basic particles inside the atoms
The electrons themselves were first discovered by JJ Thompson through experiments with vacuum tubes, while the electron charge was discovered by Robert Milikan.
Proton discovered Goldstein through a cathode ray experiment and Neutron by James Chadwick

subatomic particles in an atom
the three particles of an atom
neutrons
the mass, charge, and location of protons, neutrons, and electrons in atoms
Keywords: protons, neutrons, electrons, subatomic particles, isotope, coulomb































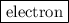
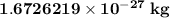 . The mass of electron is approximately 1/1836 of the mass of the proton. So the mass of electron is
. The mass of electron is approximately 1/1836 of the mass of the proton. So the mass of electron is 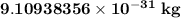 . But the mass of neutron is
. But the mass of neutron is 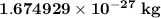 which is nearly equal to that of a proton. So electrons have the least mass among all the subatomic particles.
which is nearly equal to that of a proton. So electrons have the least mass among all the subatomic particles.

