
Chemistry, 13.03.2021 04:00 youngcie04
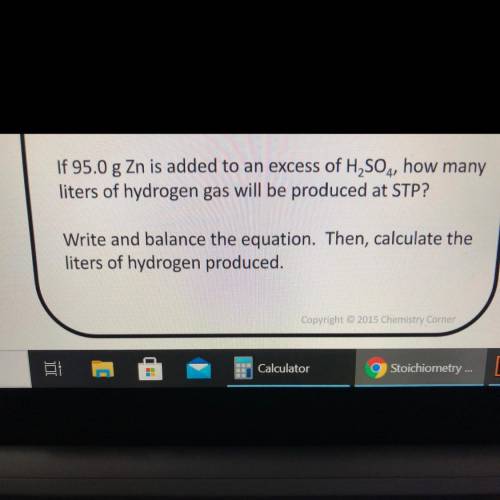
If 95.0 g Zn is added to an excess of H2SO4, how many
liters of hydrogen gas will be produced at STP?
Write and balance the equation. Then, calculate the
liters of hydrogen produced.
Please help

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1

Chemistry, 22.06.2019 13:00
In a copper wire, a temperature increase is the result of which of the following
Answers: 1

Chemistry, 22.06.2019 13:30
Why does asexual reproduction result in offspring with identicle genetic variation
Answers: 2

Chemistry, 22.06.2019 19:30
Describe the forces both attractive and repulsive that occur as two atoms move closer together.
Answers: 1
You know the right answer?
If 95.0 g Zn is added to an excess of H2SO4, how many
liters of hydrogen gas will be produced at ST...
Questions




Mathematics, 21.07.2019 06:00




Mathematics, 21.07.2019 06:00




History, 21.07.2019 06:00




Mathematics, 21.07.2019 06:00



Business, 21.07.2019 06:00



