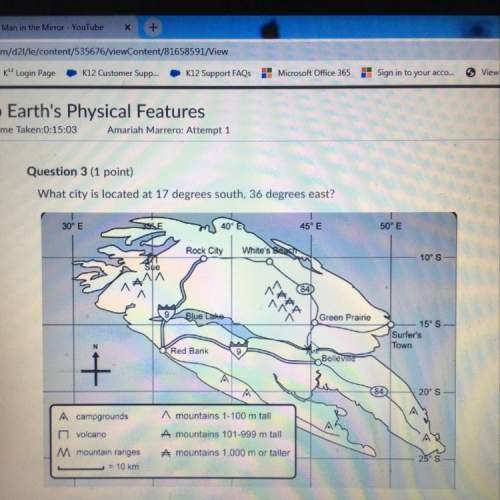
(d) A compound contains carbon and hydrogen only. On combustion, 0.150 g of
compound gives 0.488 g CO2 and 0.150 g H20. The molar mass is found to
52.09.
0) What is the mass percentage of each element in the compound?
(i) Calculate the empirical formula of the compound.
(iii) Calculate the molecular formula of the compound.
Page 2 of 8

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 00:30
Maya wrote if you step to describe how carbon circulates between the atmosphere and living organisms
Answers: 1

Chemistry, 23.06.2019 01:00
Which fossil fuel is mainly used for heating and cooking? a. electricity b. coal c. petroleum d. natural gas
Answers: 2

Chemistry, 23.06.2019 03:30
Astudent uses universal ph paper to find the ph of three solutions . solution a has a ph of 5 solution b has a ph of 11 and solution c has a ph of 7 identify which solution is acidic which solution is neutral and which solution is basic
Answers: 1
You know the right answer?
(d) A compound contains carbon and hydrogen only. On combustion, 0.150 g of
compound gives 0.488 g...
Questions


Mathematics, 31.10.2020 01:00

Mathematics, 31.10.2020 01:00

Mathematics, 31.10.2020 01:00


Mathematics, 31.10.2020 01:00


Mathematics, 31.10.2020 01:00


Chemistry, 31.10.2020 01:00



Mathematics, 31.10.2020 01:00



Mathematics, 31.10.2020 01:00


Physics, 31.10.2020 01:00

Computers and Technology, 31.10.2020 01:00