
Chemistry, 09.03.2021 02:50 quincyjosiah07
A chemistry student needs to standardize a fresh solution of sodium hydroxide. He carefully weighs out 32.mg of oxalic acid H2C2O4, a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in 250.mL of distilled water. The student then titrates the oxalic acid solution with his sodium hydroxide solution. When the titration reaches the equivalence point, the student finds he has used 60.9mL of sodium hydroxide solution. Calculate the molarity of the student's sodium hydroxide solution. Round your answer to 2 significant digits. M

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:40
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1

Chemistry, 22.06.2019 16:40
Identify the lewis acid in this balanced equation: ag+ + 2nh3 -> ag(nh3)2+a. ag+b. nh3c. ag(nh3)2+
Answers: 1


Chemistry, 22.06.2019 19:10
Δu of , in kj/kg, as it isto k, (a)as a of , (b) at , (c) at .
Answers: 2
You know the right answer?
A chemistry student needs to standardize a fresh solution of sodium hydroxide. He carefully weighs o...
Questions

Social Studies, 21.11.2020 14:00

Arts, 21.11.2020 14:00

Social Studies, 21.11.2020 14:00


Mathematics, 21.11.2020 14:00


English, 21.11.2020 14:00

Biology, 21.11.2020 14:00

Geography, 21.11.2020 14:00

Mathematics, 21.11.2020 14:00

History, 21.11.2020 14:00

Mathematics, 21.11.2020 14:00

English, 21.11.2020 14:00

History, 21.11.2020 14:00

Arts, 21.11.2020 14:00

Business, 21.11.2020 14:00


English, 21.11.2020 14:00

Mathematics, 21.11.2020 14:00

Mathematics, 21.11.2020 14:00

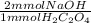 = 0.72 mmol NaOH
= 0.72 mmol NaOH

