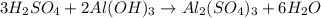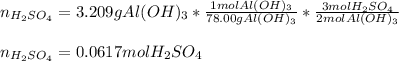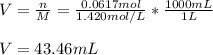Chemistry, 06.03.2021 08:00 jasminellenaee
What volume in milliliters of 1.420 M sulfuric acid is needed to neutralize 3.209 g of aluminum hydroxide 3 H 2 SO 4 (aq)+2 Al(OH) 3 (aq) Al 2 (SO 4 ) 3 (aq)+6 H 2 O

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3

Chemistry, 22.06.2019 10:30
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1

Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 23.06.2019 00:00
What conclusion can you draw from this experiment about the components of the black ink?
Answers: 3
You know the right answer?
What volume in milliliters of 1.420 M sulfuric acid is needed to neutralize 3.209 g of aluminum hydr...
Questions

History, 02.09.2020 04:01

History, 02.09.2020 04:01

Mathematics, 02.09.2020 04:01

Physics, 02.09.2020 04:01










Mathematics, 02.09.2020 04:01


Chemistry, 02.09.2020 04:01



Mathematics, 02.09.2020 04:01