
Chemistry, 05.03.2021 09:50 mrsqueenbabe516
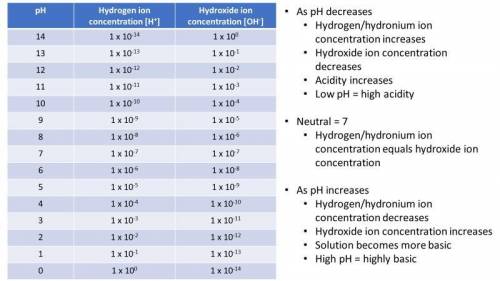
5. What is the approximate hydrogen/hydronium ion concentration in a cup of tea? (1 point)
a) What is the approximate hydroxide ion concentration in a cup of tea? (1 point)
b) Which is higher? (2 points)
(tea has a pH of 4-6)
6. What is the approximate hydrogen/hydronium ion concentration in bleach? (1 point)
a) What is the approximate hydroxide ion concentration in bleach? (1 point)
b) Which is higher? (2 points)
(bleach pH is >12)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
According to the tide table below what time of day will the highest tide occur?
Answers: 1


Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 22.06.2019 12:20
Which is an example of the practical pursuit of alchemy? a. forming perfect substances. b. transforming base metals. c. developing metalworking techniques. d. linking spiritual characteristics with material substances.
Answers: 1
You know the right answer?
5. What is the approximate hydrogen/hydronium ion concentration in a cup of tea? (1 point)
a) What...
Questions








Social Studies, 29.07.2019 05:30



Biology, 29.07.2019 05:30





Chemistry, 29.07.2019 05:30

Social Studies, 29.07.2019 05:30





