
Chemistry, 05.03.2021 04:40 aiyanaxmartinez
While performing a neutralization reaction, Jonna added 27.55 mL of 0.144 M H2SO4 to 43.84 mL of 0.316 M KOH. How many moles of OH- are unreacted in the solution after the neutralization is complete?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:30
Which is a chemical property of iron? a. it forms iron oxide (rust) when exposed to moisture and air. b. it is a gray–black metal that is hard to the touch. c. it has a melting point of 2795°f (1536°c). d. it is a good conductor of heat
Answers: 2

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1
You know the right answer?
While performing a neutralization reaction, Jonna added 27.55 mL of 0.144 M H2SO4 to 43.84 mL of 0.3...
Questions


English, 27.08.2019 05:00

English, 27.08.2019 05:00

Mathematics, 27.08.2019 05:00






English, 27.08.2019 05:00

History, 27.08.2019 05:00



Mathematics, 27.08.2019 05:00

Mathematics, 27.08.2019 05:00


Biology, 27.08.2019 05:00

History, 27.08.2019 05:00


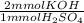 = 7.934 mmol KOH
= 7.934 mmol KOH

