
Chemistry, 05.03.2021 01:40 magicalpenguin48
What pair of elements would have the most similar properties based on their locations on the periodic table?
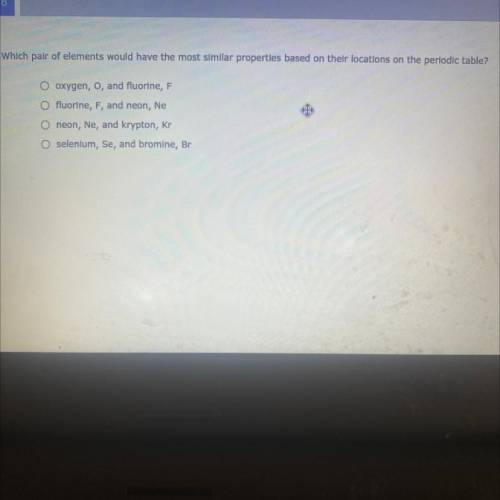

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
If a polyatomic ionic compound has gained two hydrogen ions, then how does its name begin?
Answers: 3

Chemistry, 22.06.2019 07:30
Given that 1 mi = 1760 yd, determine what conver- sion factor is appropriate to convert 1849 yd to miles; to convert 2.781 mi to yards.
Answers: 2


Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2
You know the right answer?
What pair of elements would have the most similar properties based on their locations on the periodi...
Questions





Health, 20.12.2019 08:31

Biology, 20.12.2019 08:31

Mathematics, 20.12.2019 08:31

Chemistry, 20.12.2019 08:31


Mathematics, 20.12.2019 08:31


Social Studies, 20.12.2019 08:31



Mathematics, 20.12.2019 08:31

Biology, 20.12.2019 08:31


Social Studies, 20.12.2019 08:31

Mathematics, 20.12.2019 08:31



