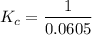Mercury(II) oxide decomposes to form mercury and oxygen, like this:
2Hg (l) + O2 (g) → 2HgO (s)
At a certain temperature, a chemist finds that a reaction vessel containing a mixture of mercury(II) oxide, mercury, and oxygen at equilibrium has the following composition:
compound amount
Hg 14.7g
O2 13.4g
HgO 17.8g
Calculate the value of the equilibrium constant for this reaction. Round your answer to significant digits. Clears your work. Undoes your last action. Provides information about entering answers.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1

Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1

Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1
You know the right answer?
Mercury(II) oxide decomposes to form mercury and oxygen, like this:
2Hg (l) + O2 (g) → 2HgO (s)
Questions



Mathematics, 24.02.2022 08:30

Physics, 24.02.2022 08:30

Chemistry, 24.02.2022 08:30

English, 24.02.2022 08:30

Computers and Technology, 24.02.2022 08:30


History, 24.02.2022 08:30



Social Studies, 24.02.2022 08:30

Mathematics, 24.02.2022 08:30



Computers and Technology, 24.02.2022 08:30

Social Studies, 24.02.2022 08:30


Biology, 24.02.2022 08:30

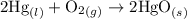
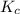 contains aqueous an dgas species only.
contains aqueous an dgas species only. ![$K_c=\frac{1}{[O_2]}$](/tpl/images/1168/7990/eab15.png) ............(1)
............(1)![$[O_2]= \frac{n_{O_2}}{V_{soln}}$](/tpl/images/1168/7990/80b5d.png) ................... (2)
................... (2)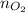 = no. of moles of oxygen gas (mol)
= no. of moles of oxygen gas (mol)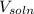 = volume of solution (L)
= volume of solution (L)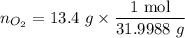
![$[O_2] =\frac{0.418 \ \text{mol}}{6.9 \ \text{L}}$](/tpl/images/1168/7990/d9674.png)