
Chemistry, 04.03.2021 19:30 michael4443
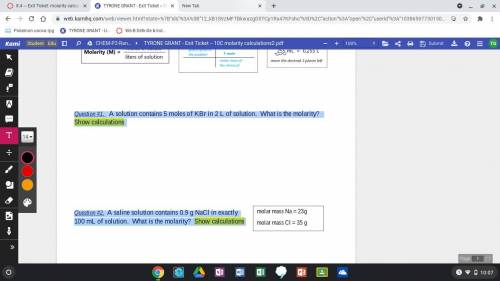
Question #1. A solution contains 5 moles of KBr in 2 L of solution. What is the molarity? Show calculations Question #2. A saline solution contains 0.9 g NaCl in exactly 100 mL of solution. What is the molarity? Show calculations

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 18:10
Given the following at 25c calculate delta hf for hcn (g) at 25c. 2nh3 (g) +3o2 (g) + 2ch4 (g) > 2hcn (g) + 6h2o (g) delta h rxn= -870.8 kj. delta hf=-80.3 kj/mol for nh3 (g), -74.6 kj/mol for ch4, and -241.8 kj/mol for h2o (g)
Answers: 1

Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1

Chemistry, 23.06.2019 01:30
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2
You know the right answer?
Question #1. A solution contains 5 moles of KBr in 2 L of solution. What is the molarity? Show calcu...
Questions



Mathematics, 13.11.2019 18:31

Biology, 13.11.2019 18:31

Mathematics, 13.11.2019 18:31




Biology, 13.11.2019 18:31

English, 13.11.2019 18:31


Social Studies, 13.11.2019 18:31

Social Studies, 13.11.2019 18:31


Chemistry, 13.11.2019 18:31





Mathematics, 13.11.2019 18:31



