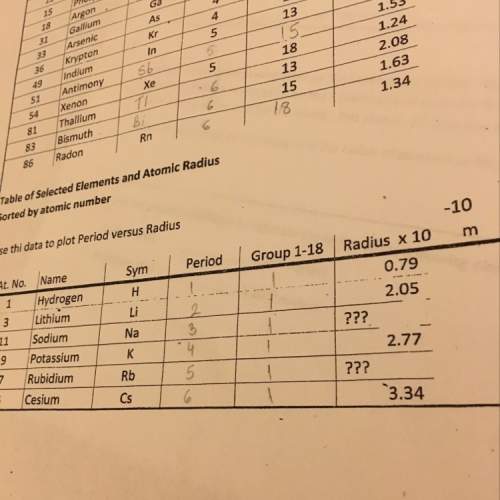
Chemistry, 03.03.2021 20:40 portedon8644
The starship enterprise uses the compound, B5H9 and oxygen gas as fuel. This is the balanced equation:
2B5H9 + 12O2 = 5B2O3 + 9H2O
If one fuel tank holds 126 g of B5H9 and the other holds 192 g of Oxygen how much B2O3 will be produced? What is the limiting reagent? How much of the excess fuel will be left over? If 171.3 g of B2)3 is actually produced, what is the percent yield?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 06:00
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be
Answers: 1

You know the right answer?
The starship enterprise uses the compound, B5H9 and oxygen gas as fuel. This is the balanced equatio...
Questions

Geography, 12.04.2021 21:50





Mathematics, 12.04.2021 21:50

Spanish, 12.04.2021 21:50

Computers and Technology, 12.04.2021 21:50

Social Studies, 12.04.2021 21:50



English, 12.04.2021 21:50


Mathematics, 12.04.2021 21:50

Mathematics, 12.04.2021 21:50




Computers and Technology, 12.04.2021 21:50