
Chemistry, 01.03.2021 22:00 guzmangisselle
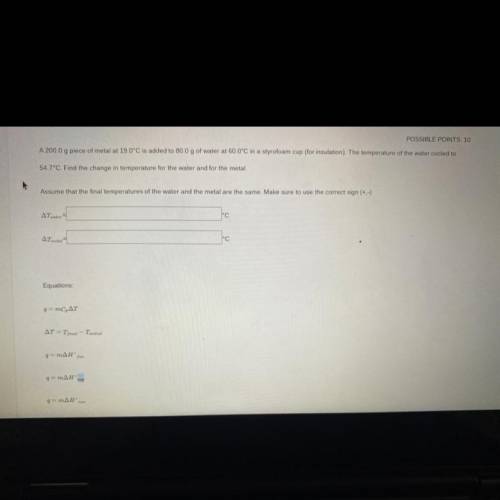
a 200g peice of metal at 19C is added to 80g of water at 60C in a styrofoam cup(for insulation). the temperature of the water is cooled at 54.7C. Find the temperature for the water and the metal. Assume that the final temperatures for the water and the metal are the same. make sure to use the correct sign

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:10
What does a particular point on a line of a phase diagram represent? o a. the maximum temperature a substance can exist at without bonds breaking b. the pressure created by the kinetic energy of molecules at a particular temperature c. the melting point or boiling point of a substance at a specific pressure d. the conditions in which temperature and pressure have equal effects on a substance
Answers: 2

Chemistry, 22.06.2019 05:30
What royal scientist used the 29th day of frozen vapor to encounter elements for mastering new culinary creations?
Answers: 1

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3
You know the right answer?
a 200g peice of metal at 19C is added to 80g of water at 60C in a styrofoam cup(for insulation). the...
Questions

Mathematics, 25.11.2021 06:40



Mathematics, 25.11.2021 06:40

Mathematics, 25.11.2021 06:40



Mathematics, 25.11.2021 06:40





History, 25.11.2021 06:40

Advanced Placement (AP), 25.11.2021 06:40



Biology, 25.11.2021 06:40



Mathematics, 25.11.2021 06:40



