
Chemistry, 01.03.2021 04:10 krystalhurst97
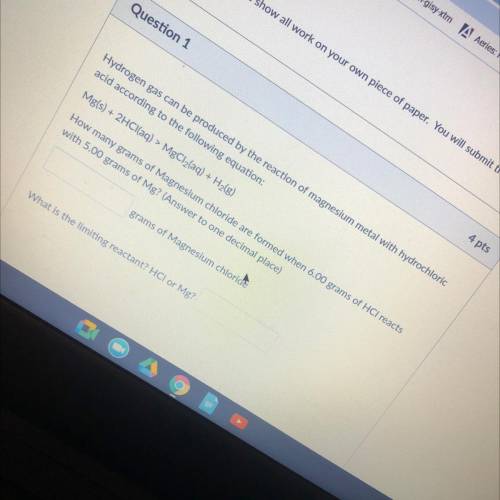
Hydrogen gas can be produced by the reaction of magnesium metal with hydrochloric
acid according to the following equation:
Mg(s) + 2HCl(aq) > MgCl2(aq) + H2(g)
How many grams of Magnesium chloride are formed when 6.00 grams of HCl reacts
with 5.00 grams of Mg? (Answer to one decimal place)
grams of Magnesium chloride
What is the limiting reactant? HCI or Mg?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 12:00
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1

Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3

Chemistry, 22.06.2019 12:30
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1
You know the right answer?
Hydrogen gas can be produced by the reaction of magnesium metal with hydrochloric
acid according to...
Questions



Health, 18.03.2021 02:00

English, 18.03.2021 02:00

Spanish, 18.03.2021 02:00




Mathematics, 18.03.2021 02:00

English, 18.03.2021 02:00


English, 18.03.2021 02:00



Mathematics, 18.03.2021 02:00

History, 18.03.2021 02:00







