TV SLABI
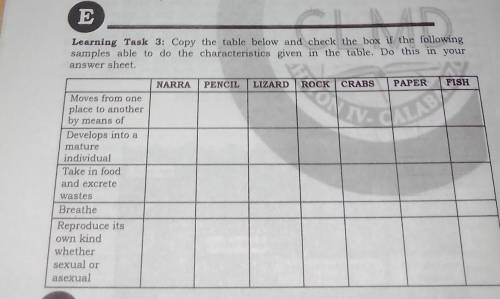
Learning Task 3: Copy the table below and check the box if the following
samples able...

Chemistry, 26.02.2021 21:30 Leeyah9587
TV SLABI
Learning Task 3: Copy the table below and check the box if the following
samples able to do the characteristics given in the table. Do this in your
answer sheet.
NARRA
PENCIL
LIZARD
ROCK
CRABS
PAPER
FISH
Moves from one place to another by means of
Develops into a mature individual
Take in food and excrete wastes
Breathe
Reproduce its
own land whether sexual or asexual
send asnswer plss :>
SCIENCE GRADE 7

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Nconcentration refers to the molar concentration of an ion in solution. it may be identical to, or greater or less than, the molar concentration of the compound containing the ion that was used to make the solution. for soluble salts, the molarity of a particular ion is equal to the molarity of that compound times the subscript for that ion. for example, 1 m of alcl3 is 1 m in al3+ and 3 m in cl−. 1 m of (nh4)2so4 is 2 m in nh4+ and 1 m in so42−. part a what is the concentration of k+ in 0.15 m of k2s? view available hint(s) nothing m m part b if cacl2 is dissolved in water, what can be said about the concentration of the ca2+ ion? view available hint(s) if is dissolved in water, what can be said about the concentration of the ion? it has the same concentration as the cl− ion. its concentration is half that of the cl− ion. its concentration is twice that of the cl− ion. its concentration is one-third that of the cl− ion. part c a scientist wants to make a solution of tribasic sodium phosphate, na3po4, for a laboratory experiment. how many grams of na3po4 will be needed to produce 550 ml of a solution that has a concentration of na+ ions of 0.700 m ? express your answer numerically in grams. view available hint(s) mass of na3po4 n a 3 p o 4 = nothing g provide feedback
Answers: 3

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 23.06.2019 00:30
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2

Chemistry, 23.06.2019 02:30
what is your question? collegechemistry 5+3 pts in november 1987, a massive iceberg broke loose from the antartic ice mass and floated free in the ocean. the chunk of ice was estimated to be 98 mi long, 25 mi wide, and 750 ft thick. a typical backyard swimming pool contains about 24,000 gallons of water. how many of these pools could you fill from the water in this iceberg? (assume the iceberg is a rectangular solid of the above dimensions and consists of water only). express answer in scientific notation.
Answers: 3
You know the right answer?
Questions

Mathematics, 18.10.2020 08:01

Mathematics, 18.10.2020 08:01



Mathematics, 18.10.2020 08:01




Mathematics, 18.10.2020 08:01


German, 18.10.2020 08:01

Biology, 18.10.2020 08:01

English, 18.10.2020 08:01


Mathematics, 18.10.2020 08:01

Biology, 18.10.2020 08:01


Advanced Placement (AP), 18.10.2020 08:01





