
Chemistry, 26.02.2021 14:00 abbygailgo674
68.3 grams of sodium hydroxide reacts with 78.3 grams of magnesium nitrate. grams of magnesium hydroxide will form from this reaction, and (compound) is the limiting reagent.
Blank 1:
Blank 2:

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:30
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2

Chemistry, 22.06.2019 22:00
All of the following are homogeneous mixtures except a) sugar dissolved in water. b) orange juice. c) coffee with cream. d) household vinegar. e) apple juice
Answers: 1

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1

Chemistry, 23.06.2019 03:50
How many moles of potassium are needed to react completely with 12.8 moles of magnessium bromide?
Answers: 2
You know the right answer?
68.3 grams of sodium hydroxide reacts with 78.3 grams of magnesium nitrate. grams of magnesium hydr...
Questions





Health, 28.02.2020 20:51


Mathematics, 28.02.2020 20:51


Biology, 28.02.2020 20:51




Chemistry, 28.02.2020 20:51


Social Studies, 28.02.2020 20:51

Biology, 28.02.2020 20:51


Mathematics, 28.02.2020 20:51


History, 28.02.2020 20:51

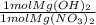 = 0.528 mol Mg(OH)₂
= 0.528 mol Mg(OH)₂ 

