
Chemistry, 25.02.2021 19:20 spowell5993
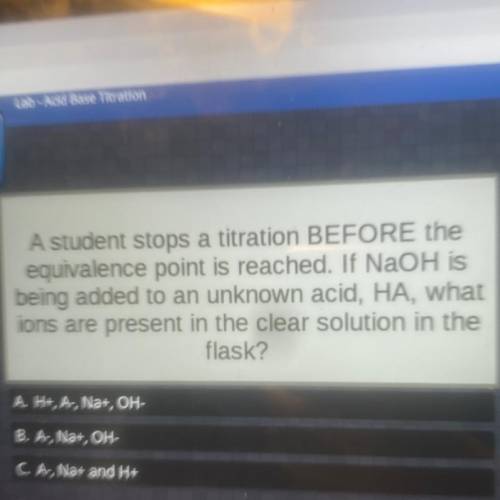
A student stops a titration BEFORE the equivalence point is reached. If NaOH is being added to an unknown acid, HA, what ions are present in the clear solution in the flask?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1


Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3
You know the right answer?
A student stops a titration BEFORE the equivalence point is reached. If NaOH is
being added to an u...
Questions


Mathematics, 17.12.2020 08:20

Mathematics, 17.12.2020 08:20

Mathematics, 17.12.2020 08:20

Mathematics, 17.12.2020 08:20

Advanced Placement (AP), 17.12.2020 08:20


Health, 17.12.2020 08:20





Mathematics, 17.12.2020 08:20

Health, 17.12.2020 08:20

English, 17.12.2020 08:20

Mathematics, 17.12.2020 08:20

Advanced Placement (AP), 17.12.2020 08:20

History, 17.12.2020 08:20


Mathematics, 17.12.2020 08:20



