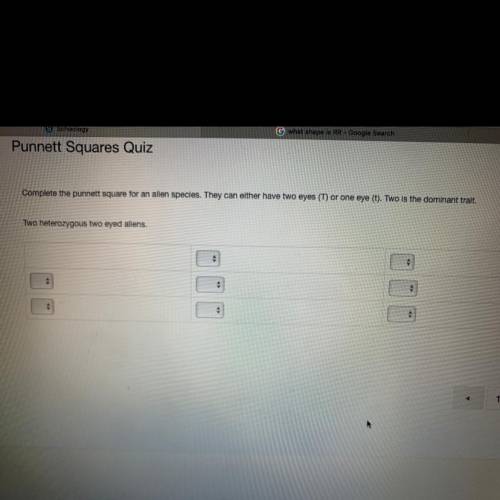
Please help asap photo attached above
...

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:10
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3

Chemistry, 22.06.2019 09:00
How are isotopes of the same chemical element alike? how are they different?
Answers: 1

Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2

Chemistry, 22.06.2019 20:20
Which symbol can be used to indicate the pressure at which a chemical reaction is carried out? 25°c 2 atm pa
Answers: 2
You know the right answer?
Questions

English, 12.10.2019 01:30


Mathematics, 12.10.2019 01:30

Social Studies, 12.10.2019 01:30

Mathematics, 12.10.2019 01:30

Mathematics, 12.10.2019 01:30

English, 12.10.2019 01:30


Mathematics, 12.10.2019 01:30


Biology, 12.10.2019 01:30




History, 12.10.2019 01:30


Mathematics, 12.10.2019 01:30