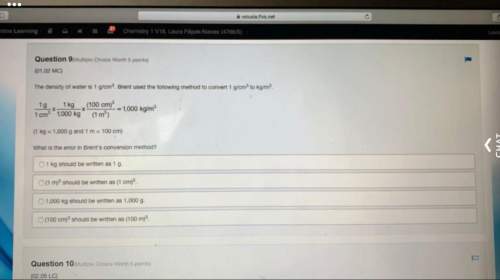
1.
How much energy (in Joules) is required to heat 300.0 g water from 23.0°C to 100.0°C?...

Chemistry, 24.02.2021 20:00 katswindle11
1.
How much energy (in Joules) is required to heat 300.0 g water from 23.0°C to 100.0°C?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 18:30
Read the claim. breakfast is an important meal. it jump starts the body’s process of using calories to break down food. appetite can decrease with age, but going too long without eating causes metabolism to slow down. current research shows that incorporating legumes such as lentils and chickpeas into meals boosts metabolism for twenty-four hours. who might benefit from this claim? people who have a fast metabolism stores that sell exercise equipment people who take vitamin supplements grocery stores that sell legumes
Answers: 1

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1
You know the right answer?
Questions







English, 11.02.2021 23:10

Social Studies, 11.02.2021 23:10


History, 11.02.2021 23:10


Mathematics, 11.02.2021 23:10

Mathematics, 11.02.2021 23:10




Mathematics, 11.02.2021 23:10

Mathematics, 11.02.2021 23:10

Mathematics, 11.02.2021 23:10

Mathematics, 11.02.2021 23:10