
Chemistry, 23.02.2021 09:00 araminaara691
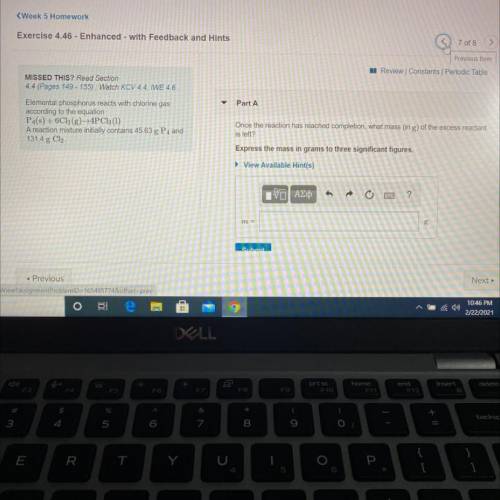
Elemental phosphorus reacts with chlorine gas
according to the equation
P4(s) + 6C12(g)+4PC13 (1)
A reaction mixture initially contains 45.63 g P4 and
131.4 g Cl.
Once the reaction has reached completion, what mass (in g) of the excess reactant is left?
Express the mass in grams to three significant figures.
please help i’ll give brainliest

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:40
Listen base your answer to the question on the information below.propane is a fuel that is sold in rigid, pressurized cylinders. most of the propane in a cylinder is liquid, with gas in the space above the liquid level. when propane is released from the cylinder, the propane leaves the cylinder as a gas. propane gas is used as a fuel by mixing it with oxygen in the air and igniting the mixture, as represented by the balanced equation below.c3h8(g) + 5o2(g) → 3co2(g) + 4h2o() + 2219.2 kja small amount of methanethiol, which has a distinct odor, is added to the propane to consumers detect a propane leak. in methanethiol, the odor is caused by the thiol functional group (–sh). methanethiol, ch3sh, has a structure that is very similar to the structure of methanol.what is the correct structural formula for a molecule of methanethiol
Answers: 3

Chemistry, 22.06.2019 18:30
Which of the following nuclei would be the least stable a 2 protons, 2 neutrons b 1 proton 1 neutron c 1 proton 3 neutrons d 1 proton 2 neutrons
Answers: 3

Chemistry, 22.06.2019 22:00
In order to complete this lab. you will need to be familiar with some common chemistry terms. complete the chemical change puzzle and list the relevant terms and their meaning below a.rectant b.product c.supernate
Answers: 3

Chemistry, 23.06.2019 02:30
What role does weathering have in shaping earth’s surface? a) it allows sediments to fall out of a medium. b) it sediments settle on a new surface. c) it breaks down older material into sediments. d) it transports sediments to a different location. will give brainliest, answer quickly.
Answers: 2
You know the right answer?
Elemental phosphorus reacts with chlorine gas
according to the equation
P4(s) + 6C12(g)+4PC13...
P4(s) + 6C12(g)+4PC13...
Questions




Chemistry, 17.12.2020 20:00

Physics, 17.12.2020 20:00

English, 17.12.2020 20:00


Mathematics, 17.12.2020 20:00

Mathematics, 17.12.2020 20:00

English, 17.12.2020 20:00

Mathematics, 17.12.2020 20:00



English, 17.12.2020 20:00

English, 17.12.2020 20:00


Chemistry, 17.12.2020 20:00


Mathematics, 17.12.2020 20:00

Arts, 17.12.2020 20:00



