

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
How much energy is made when a pice of wood burns. how do you know
Answers: 2

Chemistry, 22.06.2019 10:20
In a reaction equation, where are the products located? a.) above the arrow b.) to the right of the arrow c.) to the left of the arrow d.) below the arrow
Answers: 2

Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 23.06.2019 01:00
Imagine if during the cathode ray experiment, the size of the particles of the ray was the same as the size of the atom forming the cathode. which other model or scientific observation would have also been supported? 1. this would support dalton's postulates that proposed the atoms are indivisible because no small particles are involved. 2. this would support bohr's prediction about electrons moving in orbits having specific energy. 3. this would support bohr's prediction about electrons being randomly scattered around the nucleus in the atom. 4. this would support dalton's postulates that proposed that atoms combine in fixed whole number ratios to form compounds.
Answers: 1
You know the right answer?
The following molecular equation represents the reaction that occurs when aqueous solutions of silve...
Questions







Mathematics, 22.06.2019 14:10

Mathematics, 22.06.2019 14:20

Mathematics, 22.06.2019 14:20


History, 22.06.2019 14:20


History, 22.06.2019 14:20

English, 22.06.2019 14:20



English, 22.06.2019 14:20

Mathematics, 22.06.2019 14:20

Geography, 22.06.2019 14:20

Mathematics, 22.06.2019 14:20

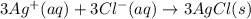

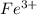 and
and  are the spectator ions.
are the spectator ions.


