
Chemistry, 22.02.2021 19:10 macimitchell
MnO4 - is a stronger oxidizing agent than ReO4 - . Both ions have charge-transfer (LMCT) bands; however, the charge-transfer band for ReO4 - is in the ultraviolet, whereas the corresponding band for MnO4 - is responsible for its intensely purple color. Are the relative positions of the charge-transfer absorptions consistent with the oxidizing abilities of these ions? Explain

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 3

Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3

Chemistry, 22.06.2019 16:30
Correct relationship between molecular formula and empirical formula
Answers: 1

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1
You know the right answer?
MnO4 - is a stronger oxidizing agent than ReO4 - . Both ions have charge-transfer (LMCT) bands; howe...
Questions

Biology, 23.05.2020 17:59




Social Studies, 23.05.2020 17:59






Mathematics, 23.05.2020 17:59

Mathematics, 23.05.2020 17:59




Chemistry, 23.05.2020 17:59


Chemistry, 23.05.2020 17:59


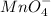 is a strong oxidizing agent.
is a strong oxidizing agent.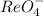 .
.

