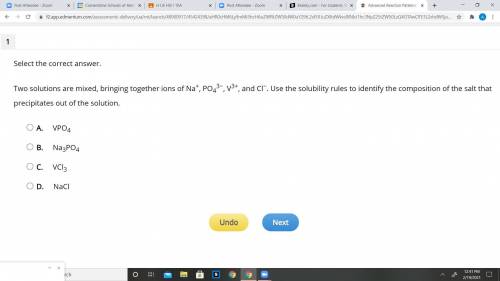
Read the Solubility Rules and answer the Question you see in the pic:
Solubility Rules
Compou...

Chemistry, 19.02.2021 22:00 solivagantjn3010
Read the Solubility Rules and answer the Question you see in the pic:
Solubility Rules
Compounds containing group 1 alkali metals or ammonium (NH4+) are soluble.
Nitrates (NO3−), chlorates (ClO3−), perchlorates (ClO4−), and acetates (C2H3O2−) are soluble.
Chlorides (Cl−), bromides (Br−), and iodides (I−) are soluble, except for compounds containing silver (Ag+), mercury(I) (Hg22+), and lead (Pb2+).
Sulfates (SO42−) are soluble, except for compounds containing calcium (Ca2+), strontium (Sr2+), barium (Ba2+), and lead (Pb2+).
Hydroxides (OH−), carbonates (CO32−), and phosphates (PO43−) are insoluble, except for compounds containing group 1 alkali metals and ammonium (NH4+).
Sulfides (S2−) are insoluble, except for compounds containing group 1 alkali metals, ammonium (NH4+), magnesium (Mg2+), calcium (Ca2+), strontium (Sr2+), and barium (Ba2+).

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Un cierto gas tiene un volumen de 800ml a 80°c y 600ml a 80°c y 600mmhg de presión. ¿cual será el volumen del gas a condiciones normales? sí el gas es oxígeno, ¿cuál será su peso? y ¿cuántas moléculas están presentes en el sistema?
Answers: 2

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 02:40
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 09:00
Ineed to find the answer of this question because i dont understand it
Answers: 1
You know the right answer?
Questions

Mathematics, 18.05.2021 05:40


Physics, 18.05.2021 05:40


Mathematics, 18.05.2021 05:40

Mathematics, 18.05.2021 05:40

Mathematics, 18.05.2021 05:40


English, 18.05.2021 05:40

Chemistry, 18.05.2021 05:40

Physics, 18.05.2021 05:40

Mathematics, 18.05.2021 05:40



Mathematics, 18.05.2021 05:40



Business, 18.05.2021 05:40


Chemistry, 18.05.2021 05:40



