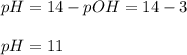Chemistry, 19.02.2021 01:00 urstruulyemily
An analytical chemist is titrating of a solution of ethylamine with a solution of . The of ethylamine is . Calculate the pH of the base solution after the chemist has added of the solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of solution added. g

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 16:10
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1

Chemistry, 22.06.2019 17:30
Observation and experimentation have led many scientists to accept a theory about the origin of the universe. this theory is called the big bang theory. scientific evidence collected and observed by scientists around the world suggests that the universe is ever expanding from a hot and dense initial state. what makes this a scientific theory? (2 points)
Answers: 2

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate
Answers: 3

Chemistry, 23.06.2019 00:20
Which diagram represents the phase tha occurs after a solid melts?
Answers: 1
You know the right answer?
An analytical chemist is titrating of a solution of ethylamine with a solution of . The of ethylamin...
Questions


History, 12.03.2020 06:58


History, 12.03.2020 06:58

History, 12.03.2020 06:58

Mathematics, 12.03.2020 06:58





Mathematics, 12.03.2020 06:59

Mathematics, 12.03.2020 06:59

English, 12.03.2020 06:59


Physics, 12.03.2020 06:59

Physics, 12.03.2020 06:59



Mathematics, 12.03.2020 06:59

History, 12.03.2020 06:59

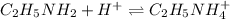
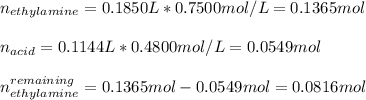
![[ethylamine]=\frac{0.0816mol}{0.1850L+0.1144L}=0.2725M](/tpl/images/1129/1639/9c401.png)
![[salt]=\frac{0.0549mol}{0.1850L+0.1144L}=0.1834M](/tpl/images/1129/1639/c810c.png)
![pOH=pKb+log(\frac{[salt]}{[base]} )\\\\pOH=3.19+log(\frac{0.1834M}{0.2725M})\\\\pOH=3.0](/tpl/images/1129/1639/77c7e.png)