Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:00
What is the empirical formula of vanadium 1 oxide given that 20.38 grams of vandium combines with oxygen to form 23.58 grams of the oxide
Answers: 1

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2


Chemistry, 22.06.2019 14:30
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2
You know the right answer?
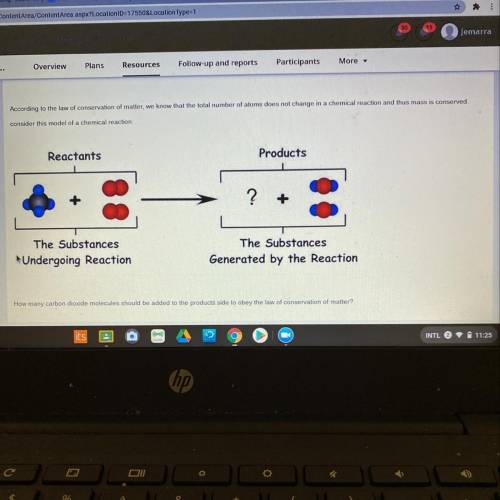
How many carbon dioxide molecules should be added to the products side to obey the law of conservati...
Questions

Chemistry, 05.11.2020 21:10


Mathematics, 05.11.2020 21:10



History, 05.11.2020 21:10

Mathematics, 05.11.2020 21:10


Mathematics, 05.11.2020 21:10


Chemistry, 05.11.2020 21:10





Mathematics, 05.11.2020 21:10

Spanish, 05.11.2020 21:10


Health, 05.11.2020 21:10

History, 05.11.2020 21:10