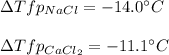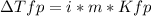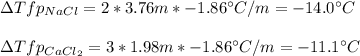Chemistry, 15.02.2021 20:10 battlemarshmell
Sodium chloride (NaCl) is commonly used to melt ice on roads during the winter. Calcium chloride (CaCl2) is sometimes used for this purpose too. Let us compare the effectiveness of equal masses of these two compounds in lowering the freezing point of water by calculating the freezing point depression of solutions containing 220. g of each salt in 1.00 kg of water. (An advantage of is that it acts more quickly because it is hygroscopic, that is, it absorbs moisture from the air to create a solution and begin the process. A disadvantage is that this compound is more costly.) Assume full dissociation of ionic compounds. Kfp(H2O)= -1.86 °C/m.
ΔTfp= °C for NaCl
ΔTfp= °C for CaCl2

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 23.06.2019 00:00
This statement about matter and its behavior is best classified as a
Answers: 1

Chemistry, 23.06.2019 00:00
What is the empirical formula of a compound that is 50.7% antimony and 49.3% selenium ?
Answers: 2
You know the right answer?
Sodium chloride (NaCl) is commonly used to melt ice on roads during the winter. Calcium chloride (Ca...
Questions

Mathematics, 26.03.2020 22:24


English, 26.03.2020 22:24


English, 26.03.2020 22:24

Mathematics, 26.03.2020 22:24


Computers and Technology, 26.03.2020 22:24

Chemistry, 26.03.2020 22:24


Health, 26.03.2020 22:24

Mathematics, 26.03.2020 22:24

Mathematics, 26.03.2020 22:24



Mathematics, 26.03.2020 22:24

History, 26.03.2020 22:24

Mathematics, 26.03.2020 22:25


Biology, 26.03.2020 22:25