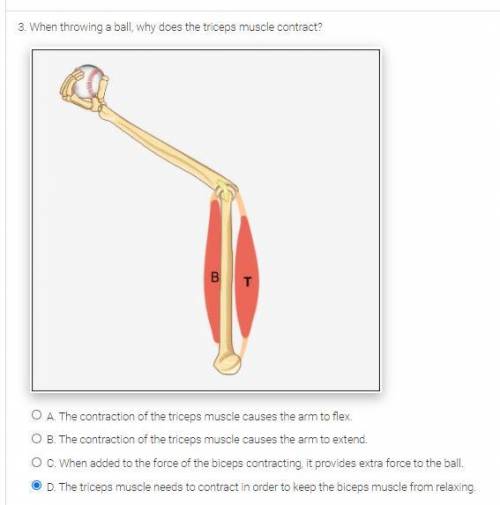
PLEASE SOMEONE ACTUALLY HELP ME NO ONES HELPING ME :((
...


Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1

Chemistry, 22.06.2019 16:50
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1

Chemistry, 23.06.2019 00:20
What type of context clue you understand the meaning of quandary?
Answers: 3
You know the right answer?
Questions



History, 25.07.2019 02:40

Biology, 25.07.2019 02:40

Social Studies, 25.07.2019 02:40

Biology, 25.07.2019 02:40


Computers and Technology, 25.07.2019 02:40


Biology, 25.07.2019 02:40




Biology, 25.07.2019 02:40

Social Studies, 25.07.2019 02:40

Biology, 25.07.2019 02:40

Biology, 25.07.2019 02:40

History, 25.07.2019 02:40