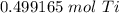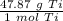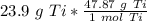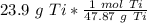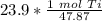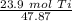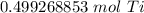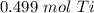Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 19:10
Δu of , in kj/kg, as it isto k, (a)as a of , (b) at , (c) at .
Answers: 2

You know the right answer?
How many moles are in 23.9 grams of titanium...
Questions










Mathematics, 14.04.2020 23:19

Mathematics, 14.04.2020 23:19


History, 14.04.2020 23:19







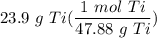 [Dimensional Analysis] Multiply/Divide [Cancel out units]:
[Dimensional Analysis] Multiply/Divide [Cancel out units]: