
Chemistry, 14.02.2021 01:20 NateTheBeast12
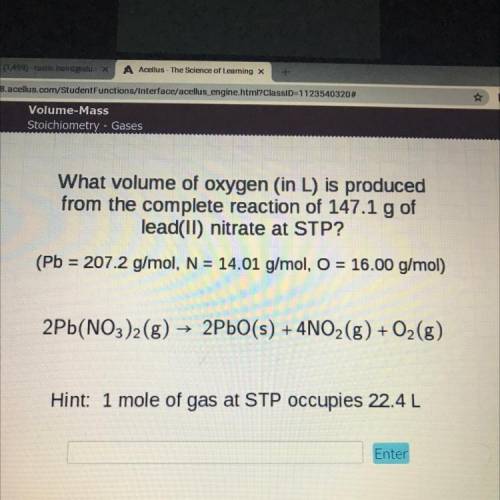
What volume of oxygen (in L) is produced
from the complete reaction of 147.1 g of
lead(II) nitrate at STP?
(Pb = 207.2 g/mol, N = 14.01 g/mol, O = 16.00 g/mol)
2Pb(NO3)2(g) → 2PbO(s) + 4NO2(g) + O2(g)
Hint: 1 mole of gas at STP occupies 22.4L

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:30
Asample of silver (with work function ? = 4.52 ev) is exposed to an ultraviolet light source (? = 200 nm), which results in the ejection of photoelectrons. what changes will be observed if: silver is replaced with copper (? = 5.10 ev) more photoelectrons ejected no photoelectrons are emitted fewer photoelectrons ejected more energetic photoelectrons (on average) less energetic photoelectrons (on average)
Answers: 3


Chemistry, 22.06.2019 02:30
Needthe meter is the standard unit for: 1) height 2) length 3) weight 4) mass
Answers: 3

You know the right answer?
What volume of oxygen (in L) is produced
from the complete reaction of 147.1 g of
lead(II) ni...
lead(II) ni...
Questions

Biology, 27.11.2019 06:31




English, 27.11.2019 06:31

English, 27.11.2019 06:31


English, 27.11.2019 06:31





Spanish, 27.11.2019 06:31









