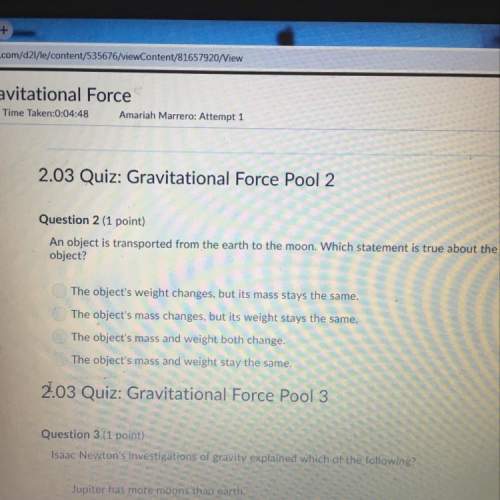
Given the following balanced chemical equation:
N2(g) + 3H2(g) + 2NH3(8)
What is the maximum...

Chemistry, 13.02.2021 07:40 terrysizemore666
Given the following balanced chemical equation:
N2(g) + 3H2(g) + 2NH3(8)
What is the maximum amount of NH3(g) that can be produced from 2.0 mol H2(g)? Assume that N2(g) is the excess reactant.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3


Chemistry, 23.06.2019 12:30
)a children’s liquid cold medicine has a density of 1.23 g/ml. if a child is to take 2.5 tsp in a dose, what is the mass in grams of this dose? (1 tsp = 5 ml)
Answers: 1
You know the right answer?
Questions

Mathematics, 24.02.2021 21:50


History, 24.02.2021 21:50


Chemistry, 24.02.2021 21:50



Mathematics, 24.02.2021 21:50

English, 24.02.2021 21:50






Mathematics, 24.02.2021 21:50

Mathematics, 24.02.2021 21:50

History, 24.02.2021 21:50

Mathematics, 24.02.2021 21:50

Mathematics, 24.02.2021 21:50

Social Studies, 24.02.2021 21:50