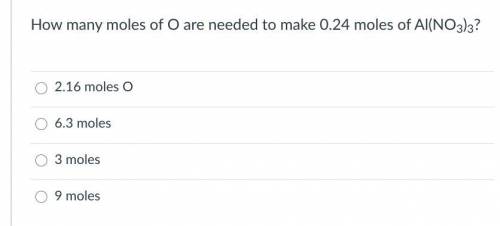
How many moles of O are needed to make 0.24 moles of Al(NO3)3?
...


Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 03:30
What is the relationship of air masses and the temperature of oceans?
Answers: 1


Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
You know the right answer?
Questions

Mathematics, 05.04.2021 06:10

Mathematics, 05.04.2021 06:10




Mathematics, 05.04.2021 06:10

Mathematics, 05.04.2021 06:10


History, 05.04.2021 06:10



Mathematics, 05.04.2021 06:10

Mathematics, 05.04.2021 06:10




Mathematics, 05.04.2021 06:10

Mathematics, 05.04.2021 06:10