Chemistry, 12.02.2021 03:30 imknutson962
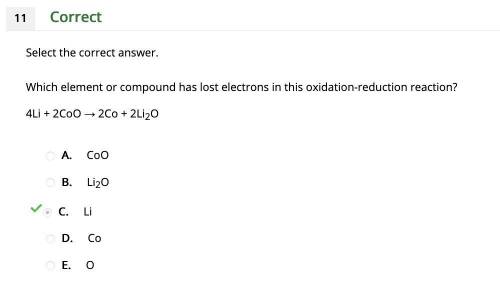
Which element or compound has lost electrons in this oxidation-reduction reaction?
4Li + 2CoO → 2Co + 2Li2O
A.
CoO
B.
Li2O
C.
Li
D.
Co
E.
O

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Bohr's model could only explain the spectra of which type of atoms? single atoms with one electron single atoms with more than one electron bonded atoms with one electron bonded atoms with more than one electron
Answers: 2


Chemistry, 22.06.2019 06:00
An atom of lithium (li) and an atom of chlorine (cl) engage in a chemical reaction. which correctly describes the structure of the resulting chemical compound? hint: consider the class of each element. the chemical compound will have a network structure. the chemical compound will have triple bonds. the chemical compound will have a ball-and-stick structure. the chemical compound will have double bonds.
Answers: 2

Chemistry, 22.06.2019 20:00
Listenbase your answer to the question on the information below.nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body.cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment.which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2
You know the right answer?
Which element or compound has lost electrons in this oxidation-reduction reaction?
4Li + 2CoO → 2Co...
Questions

Mathematics, 20.01.2021 14:00


Mathematics, 20.01.2021 14:00

Mathematics, 20.01.2021 14:00




Mathematics, 20.01.2021 14:00

Law, 20.01.2021 14:00




Mathematics, 20.01.2021 14:00


Chemistry, 20.01.2021 14:00

Mathematics, 20.01.2021 14:00




Law, 20.01.2021 14:00