
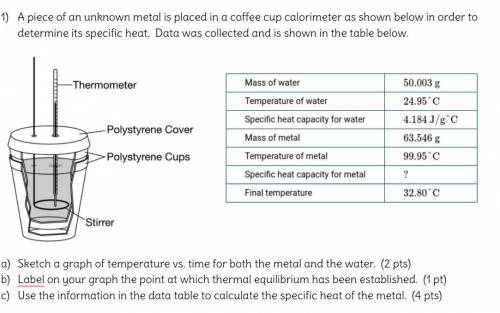
A piece of an unknown metal is placed in a coffee cup calorimeter as shown below in order to determine its specific heat. Data was collected and is shown in the table below.
Sketch a graph of temperature vs. time for both the metal and the water.
Label on your graph the point at which thermal equilibrium has been established.
Use the information in the data table to calculate the specific heat of the metal.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Which of the following is a compound? a.carbon b.oxygen c.hydrogen d.water
Answers: 2

Chemistry, 22.06.2019 02:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1


Chemistry, 22.06.2019 08:00
If 90.0 grams of ethane reacted with excess chlorine,how many grams of dicarbon hexachloride would form
Answers: 1
You know the right answer?
A piece of an unknown metal is placed in a coffee cup calorimeter as shown below in order to determi...
Questions



Mathematics, 18.08.2019 00:20





Health, 18.08.2019 00:20








Physics, 18.08.2019 00:20



History, 18.08.2019 01:10



