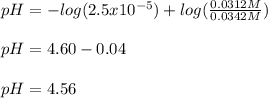Use the Henderson-Hasselbalch equation, eq. (3), to calculate the pH expected for a buffer solution prepared from this acid and its conjugate base, assuming: the mass of the weak acid is 0.2 g and the mass of the conjugate base is 0.2 g will be added to 25 mL of water. Also assume the molar mass of the weak acid is 234 and its conjugate base is the sodium salt.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:20
What is the strongest intermolecular force between an nacl unit and an h2o molecule together in a solution? covalent bonding dipole-dipole force hydrogen bonding ion-dipole force
Answers: 1

Chemistry, 22.06.2019 02:00
Which of the following is not a good technique for managing used oil? a) have specific, labeled catch pans available for technicians who are collecting oil b) spills in your shop and any releases on pavement or outside should be poured down a drain c) do not use oil containers for antifreeze or other non-similar fluids d) be prepared for oil spills with the proper absorbents
Answers: 1

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1
You know the right answer?
Use the Henderson-Hasselbalch equation, eq. (3), to calculate the pH expected for a buffer solution...
Questions


Mathematics, 27.04.2021 18:00


Mathematics, 27.04.2021 18:00

Arts, 27.04.2021 18:00

English, 27.04.2021 18:00

Mathematics, 27.04.2021 18:00

Social Studies, 27.04.2021 18:00




Mathematics, 27.04.2021 18:00



Mathematics, 27.04.2021 18:00

Mathematics, 27.04.2021 18:00

Mathematics, 27.04.2021 18:00



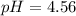
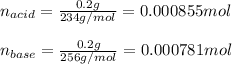
![[acid]=0.000855mol/0.025L=0.0342M](/tpl/images/1113/0020/c5975.png)
![[base]=0.000781mol/0.025L=0.0312M](/tpl/images/1113/0020/790f7.png)