
Chemistry, 11.02.2021 01:40 hihudgins902
The reaction heated up 100 g of water in the beaker from 25 °C to 50.0 °C. The specific heat capacity of water is 4.184 J/g°C. Calculate the amount of heat energy that was released by the reaction.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 05:00
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2

Chemistry, 22.06.2019 12:00
What is the subscript for oxygen in its molecular formula
Answers: 1

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1
You know the right answer?
The reaction heated up 100 g of water in the beaker from 25 °C to 50.0 °C. The specific heat capacit...
Questions



Social Studies, 29.10.2019 15:31

Mathematics, 29.10.2019 15:31






Mathematics, 29.10.2019 15:31





Mathematics, 29.10.2019 15:31

Computers and Technology, 29.10.2019 15:31

Mathematics, 29.10.2019 15:31


History, 29.10.2019 15:31

Mathematics, 29.10.2019 15:31

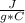 m= 100 gΔT= Tfinal - Tinitial= 50 C - 25 C= 25 C
m= 100 gΔT= Tfinal - Tinitial= 50 C - 25 C= 25 C

