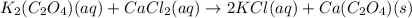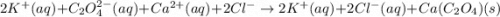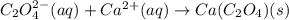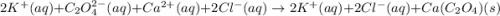Chemistry, 10.02.2021 14:00 gaceves6177
Write a balanced molecular, ionic, and net ionic equation for the following reaction. Assume the reaction occurs in
aqueous solution.
K2(C2O4)(aq)+CaCl2(aq) to
2KCl(aq)+ Ca(C2O4)(s)
balanced?
Ionic Equation:
Net Ionic Equation:

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:00
How many moles of magnesium is 3.01 x10^22 atoms of magnesium?
Answers: 1

Chemistry, 23.06.2019 01:00
Substance 33°f 100°f peanut oil solid liquid margarine solid liquid chocolate chips solid liquid which conclusion fits the data in the table? a. heat chemically changes chocolate and margarine. b. all solids become liquid at 100°f. c. removing heat from a substance it to melt. d. matter may change shape when it is heated.
Answers: 1

Chemistry, 23.06.2019 05:00
Which characteristics affect ocean water’s temperature? check all that apply. depth location mass salinity waves
Answers: 1

Chemistry, 23.06.2019 10:00
Abike ride event is 30 miles. a first aid tent is put at the 3/4 mark of the course. how many miles from the starting point is the first aid tent?
Answers: 1
You know the right answer?
Write a balanced molecular, ionic, and net ionic equation for the following reaction. Assume the rea...
Questions

Mathematics, 28.08.2020 02:01

Mathematics, 28.08.2020 02:01



Mathematics, 28.08.2020 02:01



Mathematics, 28.08.2020 02:01

Mathematics, 28.08.2020 02:01

Mathematics, 28.08.2020 02:01


Mathematics, 28.08.2020 02:01

Mathematics, 28.08.2020 02:01

World Languages, 28.08.2020 02:01

History, 28.08.2020 02:01