
Consider the following unknown solution analysis: Flame test: red flame Solutions reactions: ammonium carbonate - white precipitate (ppt.) ammonium phosphate - white ppt. Halide test: purple hexane layer The complete chemical name for the unknown compound is. The correct chemical formula of the compound is .

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:10
How many grams of naoh are needed to make 0.250 liter of a 0.500 m solution of naoh? 0.125 g 5.00 g 2.00 g
Answers: 1

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 23.06.2019 04:00
What is the volume of 2.5 moles of nitrogen gas (n2)at standard temperature and pressure (stp)?
Answers: 1

Chemistry, 23.06.2019 06:00
Complete the sentences to best explain the ranking.match the words below to the appropriate blanks in the sentences.a less polar bondhigher molar massion-dipole forcesstronger intermolecular forcesdipole-dipole forcesdispersion forceshydrogen bonding1. h2s and h2se exhibit the following intermolecular forces:.2. therefore, when comparing h2s and h2se the one with a has a higher boiling point .3. the strongest intermolecular force exhibited by h2o is . therefore, when comparing h2se and h2o the one with has a higher boiling point.
Answers: 1
You know the right answer?
Consider the following unknown solution analysis:
Flame test: red flame Solutions reactions: ammoni...
Questions

Mathematics, 26.06.2019 13:00




Mathematics, 26.06.2019 13:00


Mathematics, 26.06.2019 13:00


Geography, 26.06.2019 13:00

Physics, 26.06.2019 13:00

History, 26.06.2019 13:00

History, 26.06.2019 13:00


Mathematics, 26.06.2019 13:00



Social Studies, 26.06.2019 13:00

English, 26.06.2019 13:00


Mathematics, 26.06.2019 13:00

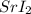 ".
".

