
Chemistry, 08.02.2021 19:10 vondah4014
Sam records the mass of his evaporating dish as 6.488 g. He records the mass of the evaporating dish and the sample of hydrate as 19.72 g. After heating the sample in the evaporating dish to constant weight, the mass of them combined is 11.344 g. How many moles of water were removed from the sample by the heating process?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3


Chemistry, 23.06.2019 01:30
Select the correct answer from each drop-down menu. to make a table of the elements, dmitri mendeleev sorted the elements according to their . he then split the list of elements into several columns so that elements beside each other had similar .
Answers: 2
You know the right answer?
Sam records the mass of his evaporating dish as 6.488 g. He records the mass of the evaporating dish...
Questions

English, 06.04.2021 19:10

Chemistry, 06.04.2021 19:10

English, 06.04.2021 19:10



Mathematics, 06.04.2021 19:10


Mathematics, 06.04.2021 19:10


Mathematics, 06.04.2021 19:10

Mathematics, 06.04.2021 19:10


Mathematics, 06.04.2021 19:10





Mathematics, 06.04.2021 19:10

English, 06.04.2021 19:10

Mathematics, 06.04.2021 19:10

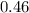 moles of water were removed from the sample by the heating process
moles of water were removed from the sample by the heating process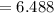 grams
grams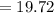 grams
grams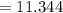 grams
grams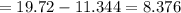 grams
grams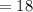 grams
grams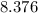 Grams of water is equal to
Grams of water is equal to 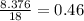 moles
moles

