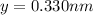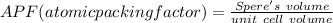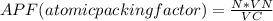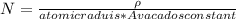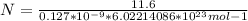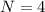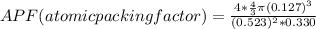Chemistry, 06.02.2021 05:40 91miketaylor
Consider a hypothetical metal that has a density of 11.6 g/cm3, an atomic weight of 157.6 g/mol, and an atomic radius of 0.127 nm. Compute the atomic packing factor if the unit cell has tetragonal symmetry; values for the and lattice parameters are 0.523 nm and 0.330 nm, respectively.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 22.06.2019 21:00
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3


Chemistry, 23.06.2019 09:30
Need , hurry pls create a superhero out of the element iron, what are its powers and his sidekick ( an element that works well with iron). how was the superhero made and who discovered him
Answers: 3
You know the right answer?
Consider a hypothetical metal that has a density of 11.6 g/cm3, an atomic weight of 157.6 g/mol, and...
Questions






Mathematics, 30.06.2019 01:30

Biology, 30.06.2019 01:30

Mathematics, 30.06.2019 01:30

Mathematics, 30.06.2019 01:30

Business, 30.06.2019 01:30

Social Studies, 30.06.2019 01:30


Social Studies, 30.06.2019 01:30

Biology, 30.06.2019 01:30

Mathematics, 30.06.2019 01:30

Mathematics, 30.06.2019 01:30

Biology, 30.06.2019 01:30

History, 30.06.2019 01:30

Social Studies, 30.06.2019 01:30

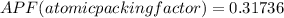
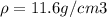
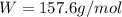
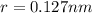
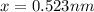 and
and