Chemistry, 06.02.2021 02:00 sarah19Nursing
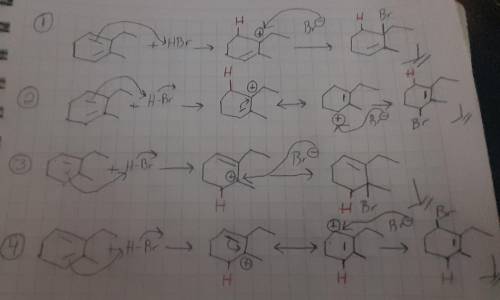
Draw all four products obtained when 2-ethyl-3-methyl-1,3-cyclohexadiene is treated with HBr at room temperature and show the mechanism of their formation. For the mechanism, include lone pairs and charges in your answer. Do not draw out any hydrogen explicitly. Do not use abbreviations such as Me or Ph.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1


Chemistry, 22.06.2019 12:40
Consider the directing effects of the substituents on salicylamide and predict the possible structures of the iodination products. which do you think will be the major product?
Answers: 1

Chemistry, 22.06.2019 14:30
What state of matter is ice a. liquid b. element c. solid d. gas
Answers: 1
You know the right answer?
Draw all four products obtained when 2-ethyl-3-methyl-1,3-cyclohexadiene is treated with HBr at room...
Questions


Mathematics, 06.01.2021 05:20



Biology, 06.01.2021 05:20

Arts, 06.01.2021 05:20


Social Studies, 06.01.2021 05:20


Mathematics, 06.01.2021 05:20

Computers and Technology, 06.01.2021 05:20



History, 06.01.2021 05:20

Mathematics, 06.01.2021 05:20

Mathematics, 06.01.2021 05:20

Health, 06.01.2021 05:20

English, 06.01.2021 05:20


English, 06.01.2021 05:20