
Chemistry, 05.02.2021 20:20 youngsavage10120
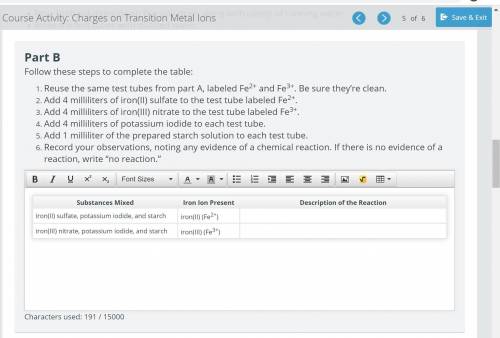
Reuse the same test tubes from part A, labeled Fe2+ and Fe3+. Be sure they’re clean.
Add 4 milliliters of iron(II) sulfate to the test tube labeled Fe2+.
Add 4 milliliters of iron(III) nitrate to the test tube labeled Fe3+.
Add 4 milliliters of potassium iodide to each test tube.
Add 1 milliliter of the prepared starch solution to each test tube.
Record your observations, noting any evidence of a chemical reaction. If there is no evidence of a reaction, write “no reaction.”

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
How are isotopes of the same chemical element alike? how are they different?
Answers: 1

Chemistry, 22.06.2019 22:00
Does the number of ions in solution increase, decrease, or remain constant? it continuously decreases. it continuously increases. it decreases at first, then increases. it increases at first, then decreases.
Answers: 3

Chemistry, 22.06.2019 23:00
What is the most common reason for matter changing its state?
Answers: 1

Chemistry, 23.06.2019 00:30
How can you write e method for the experiment of separating sand from water by filtration process? 1-materials 2-steps 3-conclusion also the same for the separating process of water and salt by filtration or distillation. quick because i need to finish my hw
Answers: 2
You know the right answer?
Reuse the same test tubes from part A, labeled Fe2+ and Fe3+. Be sure they’re clean.
Add 4 millilit...
Questions



Social Studies, 14.01.2020 19:31





History, 14.01.2020 19:31





History, 14.01.2020 19:31




Chemistry, 14.01.2020 19:31



Computers and Technology, 14.01.2020 19:31



