Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:20
Amixture of gaseous sulfur dioxide and oxygen are added to a reaction vessel and heated to 1000 k where they react to form so3(g). if the vessel contains 0.669 atm so2(g), 0.395 atm o2(g), and 0.0851 atm so3(g) after the system has reached equilibrium, what is the equilibrium constant kp for the reaction: 2 so2(g) o2(g) ⇌ 2 so3(g)
Answers: 3

Chemistry, 21.06.2019 19:00
Asyringe contains 56.05 ml of gas at 315.1 k. what volume will that gas occupy if the temperature is increased to 380.5 k? a) 12.41 b) 46.42 c) 67.68 d) 81.74
Answers: 1

Chemistry, 22.06.2019 00:00
Which of the following methods uses the decay of atomic particles in an object to find its exact age? a. fossil dating b. geologic dating c. radioactive dating d. relative dating
Answers: 1

You know the right answer?
Question Determine the concentration of hydroxide ions for a 25 °C solution with a pH of 8.22. . You...
Questions





Social Studies, 30.06.2019 11:00

Chemistry, 30.06.2019 11:00



History, 30.06.2019 11:00


English, 30.06.2019 11:00

Chemistry, 30.06.2019 11:00


History, 30.06.2019 11:00

Mathematics, 30.06.2019 11:00

History, 30.06.2019 11:00




![[OH^-]=1.7x10^{-6}M](/tpl/images/1091/8390/733f0.png)
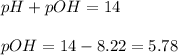
![[OH^-]=10^{-pOH}](/tpl/images/1091/8390/4817d.png)
![[OH^-]=10^{-5.78}=1.7x10^{-6}M](/tpl/images/1091/8390/28327.png)