Chemistry, 02.02.2021 04:00 ExperimentsDIYS
2. The heat from an acetylene torch is produced by burning acetylene (C2H2) in oxygen.
2C2H2 + 502 --> 4CO2 + 2H20
When 2.40mol C2H2 reacts with 7.40mol O2,
a. Which reactant is the limiting reactant?
b. How many grams of water can be produced by the reaction?
c. How many moles of the excess reactant remains?
I NEED THIS ANSWER QUICKLY IF YOU CAN ONLY DO B THATS FINE THANK YOU

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 05:00
Cucl2 + 2nano3 cu(no3)2 + 2nacl what is the percent yield of nacl if 31.0 g of cucl2 reacts with excess nano3 to produce 21.2 g of nacl? 49.7% 58.4% 63.6% 78.7%
Answers: 1

Chemistry, 22.06.2019 07:30
The volume of helium in a blimp is 6.28 x 10^9 millimeters. the density of helium in the blimp is .1786 kilogram/meter^3. find the mass of the helium in the blimp.
Answers: 1

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
You know the right answer?
2. The heat from an acetylene torch is produced by burning acetylene (C2H2) in oxygen.
2C2H2 + 502...
Questions

Mathematics, 31.03.2020 05:42

Biology, 31.03.2020 05:42

Mathematics, 31.03.2020 05:42

Computers and Technology, 31.03.2020 05:42

Mathematics, 31.03.2020 05:42

Chemistry, 31.03.2020 05:42

Mathematics, 31.03.2020 05:42


Physics, 31.03.2020 05:42

Mathematics, 31.03.2020 05:42


Social Studies, 31.03.2020 05:43


Mathematics, 31.03.2020 05:43

Mathematics, 31.03.2020 05:43




Physics, 31.03.2020 05:43

Mathematics, 31.03.2020 05:43


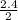 = 1.2
= 1.2 1.48
1.48