
Chemistry, 02.02.2021 02:00 oscar4hunnid
A student decreases the temperature of a 505 cm3 balloon from 465 k to 245 k. What should the new volume of the balloon be?
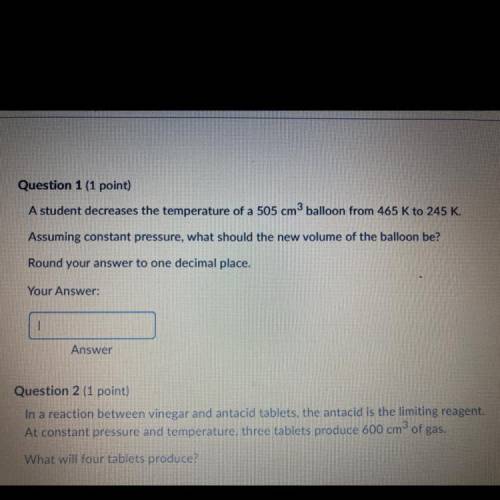

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 07:30
In the particles are arranged in a regular, repeating pattern. a)a crystalline liquid b)a crystalline solid c)all gases d)all solids
Answers: 2

Chemistry, 22.06.2019 15:30
Draw the lewis dot structure for each of the following polyatomic ions
Answers: 1

Chemistry, 23.06.2019 01:00
Imagine if during the cathode ray experiment, the size of the particles of the ray was the same as the size of the atom forming the cathode. which other model or scientific observation would have also been supported? 1. this would support dalton's postulates that proposed the atoms are indivisible because no small particles are involved. 2. this would support bohr's prediction about electrons moving in orbits having specific energy. 3. this would support bohr's prediction about electrons being randomly scattered around the nucleus in the atom. 4. this would support dalton's postulates that proposed that atoms combine in fixed whole number ratios to form compounds.
Answers: 1
You know the right answer?
A student decreases the temperature of a 505 cm3 balloon from 465 k to 245 k. What should the new vo...
Questions


History, 30.08.2019 01:30

Mathematics, 30.08.2019 01:30

Mathematics, 30.08.2019 01:30


Health, 30.08.2019 01:30



Mathematics, 30.08.2019 01:30

Social Studies, 30.08.2019 01:30


Biology, 30.08.2019 01:30

Business, 30.08.2019 01:30



History, 30.08.2019 01:30




Mathematics, 30.08.2019 01:30



