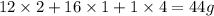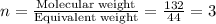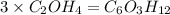Chemistry, 01.02.2021 14:00 ciaotaylor
A compound is determined to have the empirical formula C2OH4. If the molar mass of the compound is 132 g/mol, determine the molecular formula of the compound.


Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1

Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2
You know the right answer?
A compound is determined to have the empirical formula C2OH4. If the molar mass of the compound is 1...
Questions




Mathematics, 03.04.2020 05:56

Mathematics, 03.04.2020 05:56



Chemistry, 03.04.2020 05:56


Mathematics, 03.04.2020 05:56

History, 03.04.2020 05:56

Mathematics, 03.04.2020 05:56

History, 03.04.2020 05:56




Mathematics, 03.04.2020 05:56


Mathematics, 03.04.2020 05:56

Social Studies, 03.04.2020 05:56

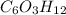
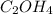 is
is