
Chemistry, 29.01.2021 19:40 adamgala3885
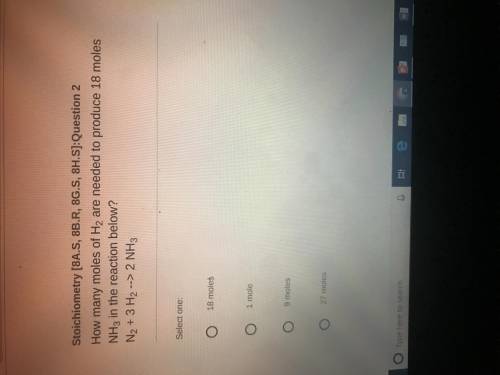
How many moles of H2 are needed to produce 18 miles NH3 in the reaction below ? N2+ 3H2 ——-> 2 NH3

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1


Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 06:00
What does it mean for something to be dissolved in watera- it is submerged in water moleculesb-it is stirred in the water moleculesc- it is surrounded by water molecules d-it has water molecules added to it
Answers: 2
You know the right answer?
How many moles of H2 are needed to produce 18 miles NH3 in the reaction below ?
N2+ 3H2 ——-> 2 N...
Questions

Mathematics, 28.06.2021 19:10



Computers and Technology, 28.06.2021 19:10

History, 28.06.2021 19:10


Chemistry, 28.06.2021 19:10





Biology, 28.06.2021 19:10

Mathematics, 28.06.2021 19:10


Mathematics, 28.06.2021 19:10

Mathematics, 28.06.2021 19:10




Biology, 28.06.2021 19:10



