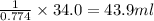Chemistry, 29.01.2021 16:30 elisakgarcia2007
The solubility in water of ionic compound X is measured and found to be 0.776g/mL at 15.°C. Calculate the volume of a saturated solution of X in water that would contain 34.0g of X at this temperature. Be sure your answer has the correct unit symbol and 3 significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
This flow chart shows the amount of energy that is emitted by each type of light. ultraviolet > blue light > yellow light > red light (maximum energy) (minimum energy) in an experiment, shining which type of light on a strip of metal would be least likely to produce the photoelectric effect? ultraviolet light dim blue light bright red light bright yellow light
Answers: 2

Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2

Chemistry, 22.06.2019 23:00
What is the measured amount of a product obtained from a chemical reaction?
Answers: 1

Chemistry, 23.06.2019 00:30
Element j is 1s 2s 2p 3s . (i) how many unpaired electrons does j have? (ii) is j a good oxidizing agent or a reducing agent? (iii) state reason for the answer.
Answers: 1
You know the right answer?
The solubility in water of ionic compound X is measured and found to be 0.776g/mL at 15.°C. Calculat...
Questions


Social Studies, 23.11.2020 14:00

Mathematics, 23.11.2020 14:00

Mathematics, 23.11.2020 14:00

History, 23.11.2020 14:00

Medicine, 23.11.2020 14:00

Biology, 23.11.2020 14:00





Computers and Technology, 23.11.2020 14:00

Mathematics, 23.11.2020 14:00

Mathematics, 23.11.2020 14:00


Computers and Technology, 23.11.2020 14:00

Mathematics, 23.11.2020 14:00



Mathematics, 23.11.2020 14:00

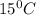 = 0.776 g/ml
= 0.776 g/ml