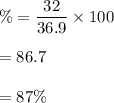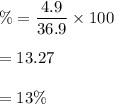Chemistry, 29.01.2021 16:30 cynayapartlow88
Large amounts of an unknown compound were isolated from seawater. The compound contained 32.0 grams ofbromine and 4.9 grams of magnesium. What is its percent composition?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 08:30
In the millikan oil drop experiment they determined that every drop had a charge which was a while number multiple of -1.60x10^-19. if a drop has a total charge of -9.60x10^-19 then how many excess electrons are contained within the drop?
Answers: 2

Chemistry, 22.06.2019 10:30
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1

Chemistry, 22.06.2019 21:30
If 22.5 of nitrogen at 748 mm hg are compressed to 725 mm hg at constant temperature. what is the new volume?
Answers: 1
You know the right answer?
Large amounts of an unknown compound were isolated from seawater. The compound contained 32.0 grams...
Questions


Mathematics, 20.09.2019 16:30

English, 20.09.2019 16:30






History, 20.09.2019 16:30


Mathematics, 20.09.2019 16:30



Mathematics, 20.09.2019 16:30


Mathematics, 20.09.2019 16:30


English, 20.09.2019 16:30

Chemistry, 20.09.2019 16:30