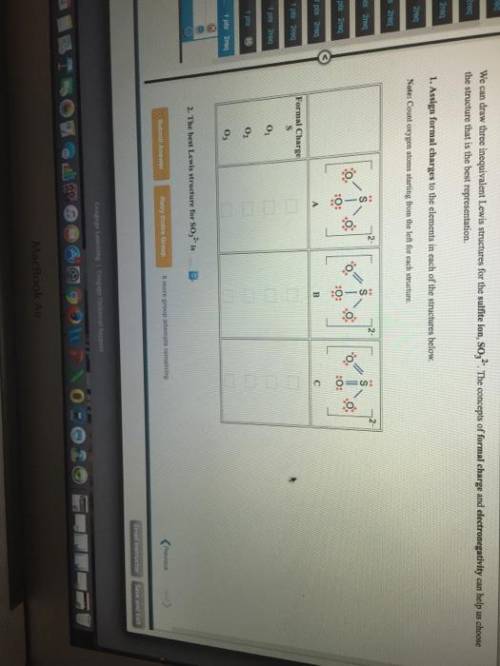
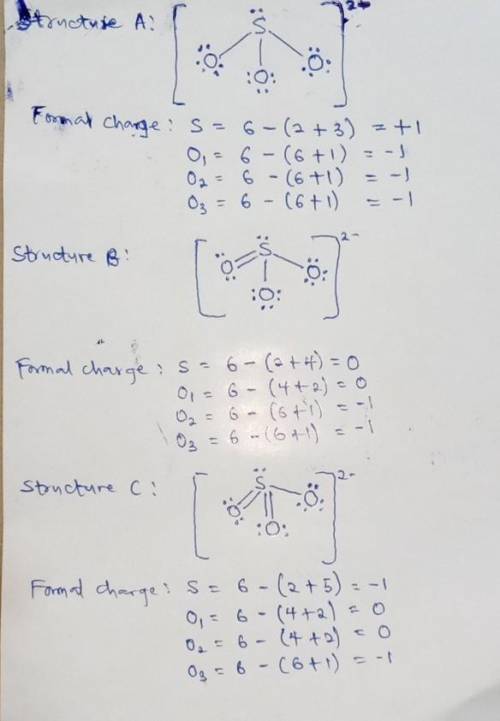
We can draw three inequivalent Lewis structures for the sulfite ion , SO32- . The concepts of formal charge and electronegativity can help us choose the structure that is the best representation. 1. Assign formal charges to the elements in each of the structures below. Note: Count oxygen atoms starting from the left for each structure. A B C Formal Charge S O1 O2 O3 2. The best Lewis structure for SO32- is

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
What term is applied to a scientist who studies ancient life, including animal and plant fossils a. anthropologist b. dendroclimatologist c. geophysicist d. paleontologist
Answers: 2

Chemistry, 22.06.2019 13:30
What produces wave a)sound b) heats c)transfer of energy d)vibrations
Answers: 2

Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1

Chemistry, 22.06.2019 17:00
Reduction is a reaction which results in a in electrons and a in positive charge of the atom or ion 1) a- loss 1) b- gain 2) a-increase 2) b-decrease
Answers: 1
You know the right answer?
We can draw three inequivalent Lewis structures for the sulfite ion , SO32- . The concepts of formal...
Questions



Mathematics, 04.05.2020 22:52

Mathematics, 04.05.2020 22:52







Mathematics, 04.05.2020 22:53

Mathematics, 04.05.2020 22:53

Mathematics, 04.05.2020 22:53


English, 04.05.2020 22:53

Mathematics, 04.05.2020 22:53


Social Studies, 04.05.2020 22:53

Mathematics, 04.05.2020 22:53

History, 04.05.2020 22:53