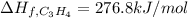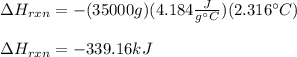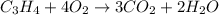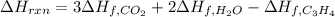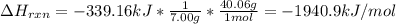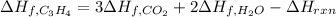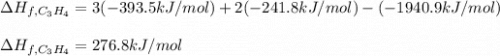Chemistry, 27.01.2021 09:40 Itsyaaboij4663
7.00 of Compound x with molecular formula C3H4 are burned in a constant-pressure calorimeter containing 35.00kg of water at 25c. The temperature of the water is observed to rise by 2.316c. (You may assume all the heat released by the reaction is absorbed by the water, and none by the calorimeter itself.) Calculate the standard heat of formation of Compound x at 25c. Be sure your answer has a unit symbol, if necessary, and round it to 3 significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 20.06.2019 18:02
How do you find a theoretical mass? is there a difference between theoretical mass and theoretical yield?
Answers: 1

Chemistry, 22.06.2019 05:00
When you mate two plants together the terms is called? answer it fast as possible plz! i have a test tomorrow!
Answers: 1


Chemistry, 22.06.2019 08:30
Which common material is an example of a polymer? (25 pts) a. steel b. plastic c. petroleum d. rubbing alcohol
Answers: 2
You know the right answer?
7.00 of Compound x with molecular formula C3H4 are burned in a constant-pressure calorimeter contain...
Questions












Mathematics, 04.04.2020 11:51

Mathematics, 04.04.2020 11:51


Mathematics, 04.04.2020 11:52

Social Studies, 04.04.2020 11:52

English, 04.04.2020 11:52