
The following reaction shows the products when sulfuric acid and aluminum hydroxide react.
2Al(OH)3 + 3H2SO4 → Al2(SO4)3 + 6H2O
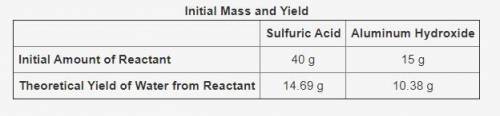
The table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory.
What is the approximate amount of the leftover reactant?
11.73 g of sulfuric acid
10.33 g of sulfuric acid
11.12 g of aluminum hydroxide
13.67 g of aluminum hydroxide

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Which other elements contain the same number of outer electrons as sodium
Answers: 3

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1


Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2
You know the right answer?
The following reaction shows the products when sulfuric acid and aluminum hydroxide react.
2Al(OH)...
Questions




History, 22.04.2020 17:18


History, 22.04.2020 17:18

Spanish, 22.04.2020 17:18

Biology, 22.04.2020 17:18


Mathematics, 22.04.2020 17:18

Mathematics, 22.04.2020 17:18





History, 22.04.2020 17:18


Mathematics, 22.04.2020 17:19




